What Is An Amu The Essential Unit Of Mass Defining Matters Fundamental Nature
What Is an Amu? The Essential Unit of Mass Defining Matters Fundamental Nature — Often overlooked in modern science, the amu (atomic mass unit) remains a cornerstone in understanding matter at the microscopic level. Defined as one-twelfth the mass of a carbon-12 atom, the amu bridges the gap between the invisible world of atoms and measurable reality.
More than just a historical relic, it shapes how physicists quantify mass, enabling precise calculations in chemistry, physics, and engineering. Far more than a unit of weight, the amu reflects the fundamental nature of mass as a measurable, constant in nature — a pillar of scientific consistency in an ever-shifting landscape of measurement.
Origin and Definition: From Carbon-12 to a Universal Benchmark
The amu traces its roots to the precise measurement of atomic masses. Early attempts to standardize mass units faltered due to inconsistencies in definitions.
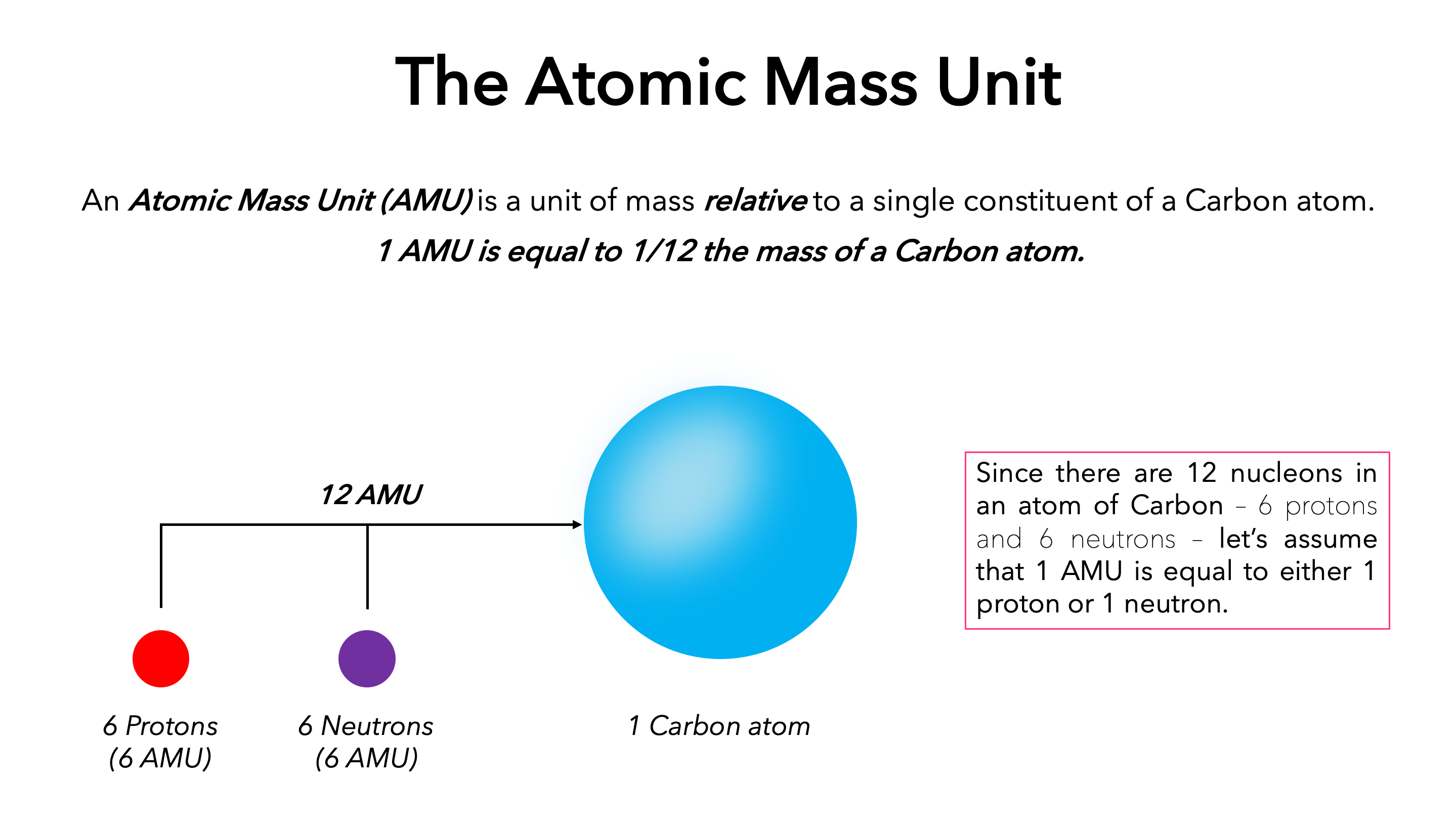
In 1961, the 18th General Conference on Weights and Measures (CGPM) anchored the amu to carbon-12 — specifically, 1/12th the mass of a naturally occurring carbon-12 atom. This choice was strategic: carbon-12 offers a stable, reproducible, and naturally abundant standard. At the time, it was recognized that mass is not merely a physical property but a foundational property tied to nature’s constants.
The carbon-12 standard allows scientists to express atomic masses with extraordinary precision—often to within a millionth of a percent—critical when modeling chemical reactions or particle behavior.
The amu was initially defined through a physical standard: a sample of pure carbon-12 constrained to a defined volume.
However, due to technological advances, modern definitions rely on exact atomic mass measurements. In 2019, the International System of Units (SI) redefined the kilogram using the fixed numerical value of the Planck constant, anchoring mass to quantum phenomena rather than physical artifacts. While the amu remains a practical unit in atomic and molecular contexts, its definition now aligns seamlessly with the quantum mechanical reality of mass, reinforcing its role in foundational physics.
As physicist Leonard I. Surd preservative noted, “The amu transforms the abstract mass of subatomic particles into a meaningful, calculable magnitude—essential for both theory and engineering.”
Why Mass Matters: The Fundamental Role of the Amu in Science
Mass is not merely a measure of weight; it is the quantifiable resistance of matter to acceleration, a property deeply embedded in the laws of physics. From Newton’s second law (F = ma) to Einstein’s relativity (E = mc²), mass determines how energy and motion interact.
The amu makes these interactions tangible at the atomic scale. A carbon-12 atom weighs roughly 12 amu, making it a natural reference point for comparing isotopes, calculating molecular weights, or analyzing nuclear reactions. For instance, the difference in mass between carbon-12 and carbon-14 — just two extra neutrons — reveals profound implications for radiometric dating and nuclear stability.
Understanding isotopic mass changes via the amu enables scientists to quantify energy release in nuclear fusion and fission, underpinning both stellar physics and power generation.
Applications Across Disciplines: From Lab benches to Industrial Scale
The amu’s influence extends far beyond theoretical physics into real-world applications.
In chemistry, it standardizes molecular formulas; knowing that glucose has a molar mass of about 180 atomic mass units (u) permits precise stoichiometric modeling. In pharmacology, drug dosing depends on accurate molecular mass calculations — enabling patient-safe delivery of therapeutic compounds.
Environmental science leverages mass precision in tracking pollutants and greenhouse gases, where parts-per-billion differences hinge on molecular weight.Materials engineering uses the amu to design alloys and composites, balancing atomic mass to achieve desired strength, conductivity, or durability. Even space exploration depends on mass accuracy—astronauts’ fuel loads, satellite orbits, and planetary mass estimates all rely on rigorously defined units like the amu.
Precision and Unity in Measurement: The Amu’s Enduring Legacy
At its core, the amu is more than a unit—it is a symbol of scientific progress.
By anchoring mass to a stable atomic standard, it unifies diverse scientific communities under a single language for measurement. This consistency enables reproducibility across laboratories, accelerates innovation in emerging technologies, and supports global scientific collaboration. As instrumentation evolves—from mass spectrometry to laser interferometry—the fundamental need for a quintessential mass unit persists.
The amu, rooted in carbon-12 and refined by quantum precision, bridges tradition and advancement.
For every atom measured, every reaction modeled, and every discovery built, the amu stands as a silent guardian of clarity.
In summary, the amu is not merely an arbitrary unit but a profound expression of how humanity quantifies the material world. Defined by nature’s constants, refined by technological insight, and deployed across countless scientific frontiers, it remains indispensable. Whether in analyzing the complexity of a protein, verifying nuclear reactions, or calibrating industrial processes, the amu mediates understanding—making the invisible measurable, the abstract precise, and the fragmented coherent.
Its name may be concise, but its impact is vast—a testament to how fundamental units shape scientific truth.



Related Post

From Soul to Sound: The Legacy of Charles Wayne Ray Charles Son

Is Courtney Still Lighting Up QVC with Fresh Flu streams and Fan Favorite Moments?

Tyla Height Unveiled: The Prodigy’s Measurements Defining a New Era inStars

Emma Heming’s Mother: The Quiet Strength Behind a Legend’s Rise

