Unlocking Life’s Blueprint: The Mechanism and Medical Power of Peptide Synthesis
Unlocking Life’s Blueprint: The Mechanism and Medical Power of Peptide Synthesis
At the frontier of biotechnology and pharmaceutical innovation, peptide synthesis stands as a cornerstone technique for crafting biological molecules with profound therapeutic potential. Guided by sophisticated chemical mechanisms, this process enables scientists to assemble amino acid sequences into functional peptides—molecules central to drug development, vaccine design, and biomedical research. Understanding the precise mechanisms driving peptide bond formation, alongside its diverse applications, reveals how synthetic peptides are transforming modern medicine.
From mimicking natural hormones to delivering targeted therapies, peptide synthesis bridges fundamental chemistry with life-saving innovation. The efficacy of this technology relies not just on theoretical principles, but on robust strategies that ensure accuracy, scalability, and biological relevance.
Peptide synthesis hinges on the formation of peptide bonds through a dehydration reaction, where the carboxyl group of one amino acid reacts with the amino group of another, releasing a water molecule and linking the two residues.
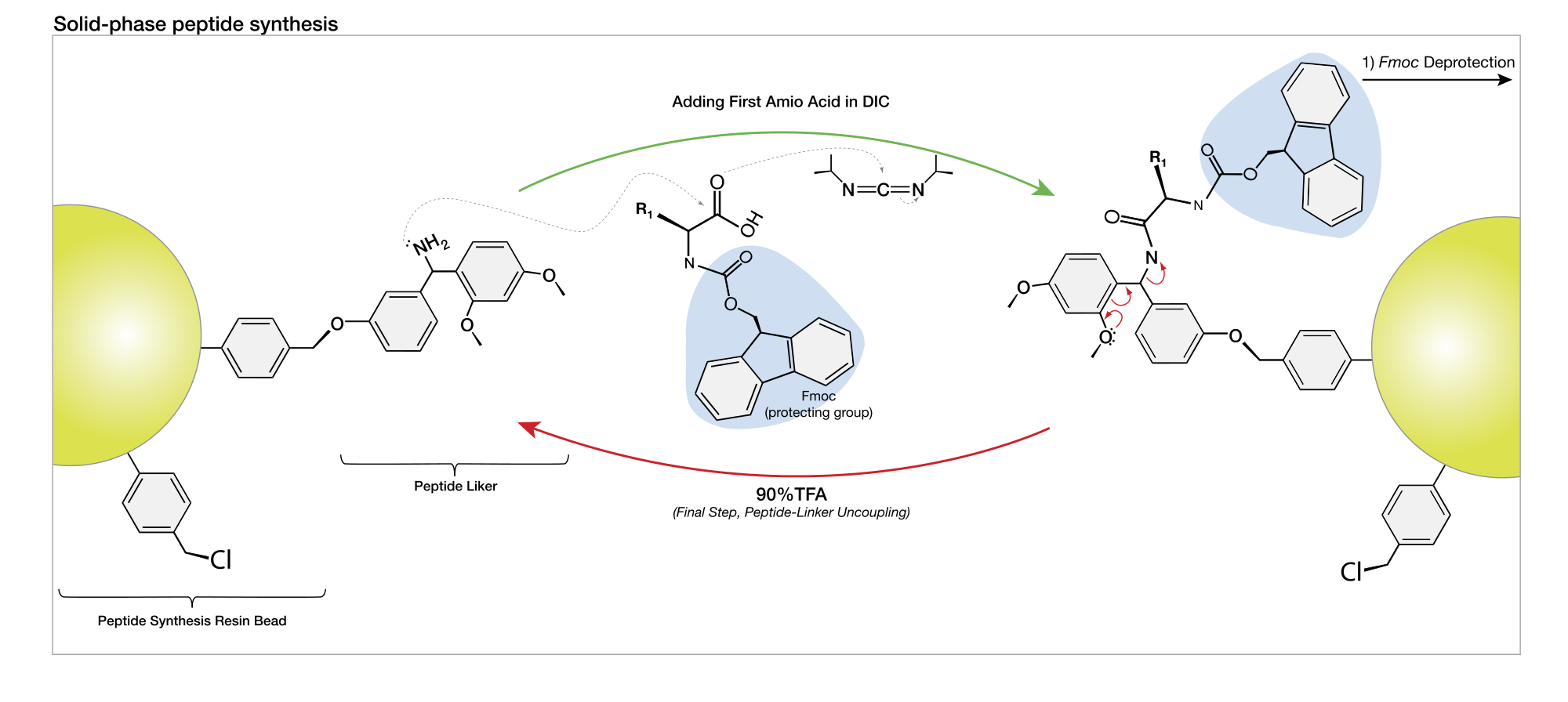
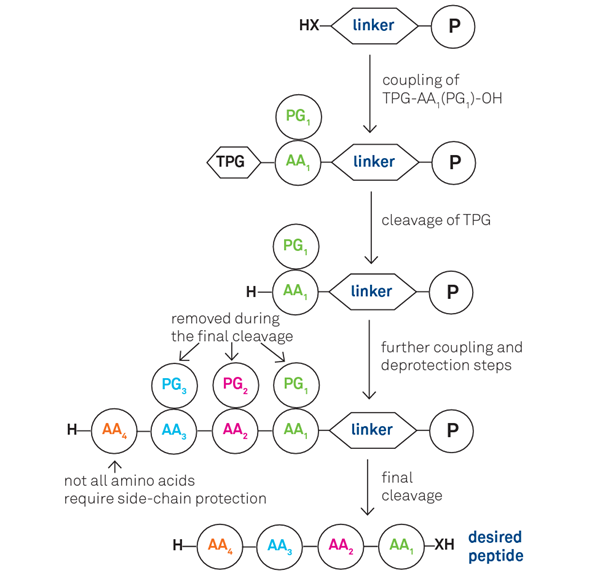
This condensation process, governed by precise mechanistic steps, occurs with high stereochemical control—specifically favoring the L-configuration of natural amino acids. In solid-phase peptide synthesis (SPPS), pioneered by Robert Bruce Merrifield in the 1960s, peptides are built sequentially on an insoluble resin support, simplifying purification and enabling automation. The method typically employs Fmoc (fluorenylmethyloxycarbonyl) or Boc (tert-butyloxycarbonyl) protecting groups to selectively mask reactive amino groups, preventing unwanted side reactions until deliberate deprotection triggers bond formation.“The elegance of solid-phase synthesis lies in its ability to streamline a complex, multistep reaction into a single, repeatable platform with minimal purification,” explains Dr.
Elena Torres, a peptide chemist at the Max Planck Institute for Medical Research.
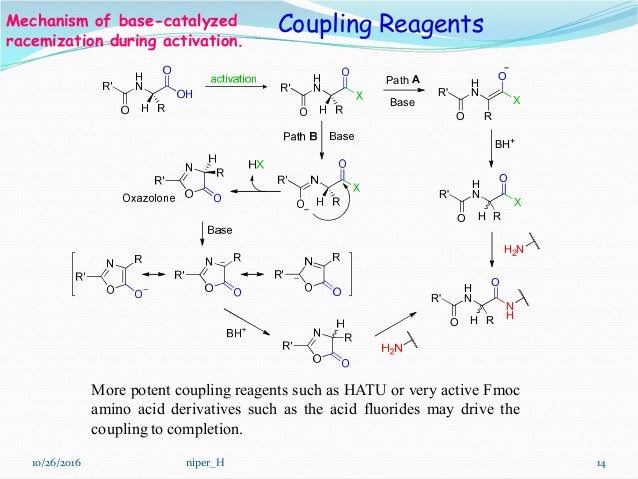
Several key stages underpin successful peptide assembly, starting with attachment to a solid support—commonly Wang resin in Fmoc-based SPPS.uitary turbine of amino acids is executed with precision: each new residue is activated using coupling reagents such as HBTU (2-(1H-Benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate) or HATU (O-(7-azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate), which enhance nucleophilic attack by the incoming amino group. Following each addition, clean-up and capping steps are critical—capping prevents deletion sequences by blocking unreacted sites, while thorough washing ensures purity.
Once the full peptide chain reaches the desired length, cleavage from the resin using strong acids like trifluoroacetic acid (TFA) also releases side-chain protecting groups, yielding the complete peptide freed as a mixture of orthogonal fragments. Finally, purification via ultra-performance liquid chromatography (UPLC) isolates the target sequence with high resolution, enabling structural confirmation and biological testing.“Each step in SPPS is a carefully orchestrated dance—timing, reagent choice, and purification certainty define the success of the final product,” notes Dr. Mehran Ekhtiari, a process chemist specializing in peptide manufacturing.
The mechanistic rigor of peptide synthesis directly enables its wide-ranging applications across medicine and research. In therapeutics, synthetic peptides serve as targeted drugs, such as dulaglutide, a glucagon-like peptide 1 (GLP-1) analog used in keeping blood glucose levels stable for type 2 diabetes. These molecules mimic natural signaling peptides with enhanced stability decay-resistant against enzymatic breakdown.
Beyond diabetes, therapeutic peptides are pivotal in oncology, delivering cytotoxic agents directly to tumor cells with minimal off-target effects, and in immunology, acting as epitope mimetics for vaccine development.“Peptides offer unmatched specificity—they can be designed to interact precisely with disease-related proteins, making them ideal candidates for next-generation biologics,” remarks Dr. Amara Nkosi, a peptide drug developer at the University of Cape Town’s biotech hub.
Beyond clinical applications, peptide synthesis fuels discovery in structural biology and drug screening.
Research teams synthesize mimetic peptides that replicate the active sites of protein complexes, enabling high-throughput assays to identify potential drug leads. In vaccine design, synthetic peptides serve as subunit vaccines, triggering immune responses without infection risks. For example, peptide-based COVID-19 vaccines encode viral spike protein fragments, training the immune system effectively and safely.“Synthesis allows rapid, customizable antigen display—critical when responding to emerging pathogens,” says Dr.
Liam Cole, senior virologist at the CDC.
Recent technological advances continue to refine synthesis efficiency and functional diversity. Automated synthesizers now handle fully automated workflows, reducing operator error and accelerating production timelines.
Parallel developments in orthogonal protecting group strategies enable the incorporation of non-natural amino acids—such as fluorescent or metallic residues—that impart new biochemical properties, expanding peptides’ utility in imaging and catalysis.“Expanding the chemical space of peptides opens doors to entirely new functionalities,” asserts Dr. Sofia Petrova, a synthetic biologist at MIT.
Despite these milestones, challenges persist.
Peptide stability in vivo remains a concern due to rapid enzymatic degradation, prompting research into cyclization and backbone modifications to prolong half-life. Additionally, ensuring correct stereochemistry and avoiding aggregation are persistent hurdles in large-scale manufacturing. Yet, ongoing innovation in solid support materials, cleavage protocols, and purification techniques steadily advances solution reliability.
Peptide synthesis is not merely a lab technique—it is a dynamic engine of biomedical progress. From its chemically intricate mechanisms to its life-saving applications, the discipline exemplifies how precision engineering at the molecular level translates into tangible health benefits. As research deepens and technology evolves, synthetic peptides will continue to illuminate new pathways in medicine, diagnostics, and targeted therapy—solidifying their role as essential tools in the life sciences.
Related Post

Joe Rogan’s Age, Height, and Net Worth Unveiled: Nationality, Single Life, and Standing in the Spotlight

Unveiling Jenn Carter’s Height: From Myths to Measurement – The Truth Behind Her Stature

Jodi Arias Nude: The Controversial Image That Ignited a Media Storm

Ally Sheedy’s Romantic Enigma: The Unlikeable Charm Behind Hollywood’s Silver Screen Muse

